I E Irodov Thermodynamics And Molecular Physics (The First Law Of Thermodynamics, Heat Capacity) Q . 2.32
I E Irodov Thermodynamics And Molecular Physics(The First Law Of Thermodynamics, Heat Capacity) Q. 2.32
The first law of thermodynamics, heat capacity is very important from the perspective to maximum score in IIT JEE. Let's take an insight look to the Weightage of Equation of gas questions is 3-4 asked every year in JEE Main exam.
So, a good study paln is key for success especially in JEE Mains. It can save your time. But It's up to you how you prepare yourself.
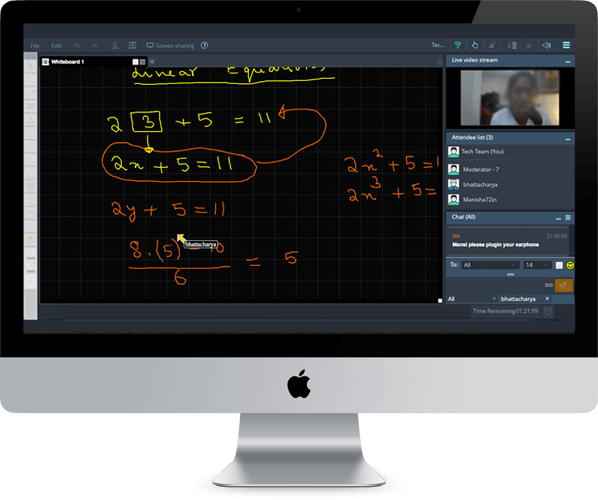
Q. 2.32 Two moles of a certain ideal gas at a temperature T0=300K were cooled isochorically so that the gas pressure reduced n=2.0 times. Then, as a result of the isobaric process, the gas expanded till its temperature got back to the initial value. Find the total amount of heat absorbed by the gas in this process.
Complete I E Irodov Thermodynamics And Molecular Physics (The First Law Of Thermodynamics, Heat Capacity) Q 2.32 Video Solution -
Indian & NRI students who wish to take admission in top engineering colleges in India, Knowing the important topics of the latest JEE Main, weightage & the first law of thermodynamics, heat capacity video solutions will help Indian & NRI students prepare faster and score more.
If any of your questions are unanswered please ask your questions below in the comment box. We will give you the solutions to your questions.
Key Point Of The First Law Of Thermodynamics, Heat Capacity:
- The primary law of thermodynamics communicates that energy is conserved, when all types of energy, including heat are taken into account.
For instance, u be the internal energy of system then delta u=energy input by heating + work done on system. This is called first law of thermodynamics. We can also say that it is the law of conversion of energy.
- Heat is the flow of energy starting with one object then onto the next caused by a distinction in temperature. C = Q/delta T. While we will regularly utilize heat capacity, heat capacities are like mass, that is their value depend on the material and on the amount of it there is.
Important 'The First Law Of Thermodynamics, Heat Capacity' Questions You Should Not Miss:
|
I E Irodov Topics Of The first Law of Thermodynamics, Heat Capacity |
Video Solutions |
|
Thermodynamics and Molecular Physics The first Law of Thermodynamics, Heat Capacity Q. 2.29 |
|
|
Thermodynamics and Molecular Physics The first Law of Thermodynamics, Heat Capacity Q. 2.35 |
|
|
Thermodynamics and Molecular Physics The first Law of Thermodynamics, Heat Capacity Q. 2.46 |
|
|
Thermodynamics and Molecular Physics The first Law of Thermodynamics, Heat Capacity Q. 2.6 |
Important Topics Questioned Covered In The First Law Of Thermodynamics, Heat Capacity:
The law of conservation of energy expresses that the complete energy of isolated system is consistent; energy can be changed starting with one structure then onto the next, but it can be neither created nor destroyed.
Following topics are covered in I E Irodov solutions:
|
I E Irodov Topics |
I E Irodov Solutions |
|
Equation of the Gas State, Processes |
|
|
Kinetic Theory of Gases. Boltzmann's Law and Maxwell's |
|
|
The Second Law of Thermodynamics. Entropy |
|
|
Liquids. Capillary Effects |
|
|
Phase Transformations |
|
|
Transport Phenomena |
Important IE Irodov Video Solutions You Should Not Miss:
- Physical Fundamentals of Mechanics
- Electrodynamics
- Oscillations and Waves
- Optics
- Atomic and Nuclear Physics
Useful JEE Main Links You Should Not Miss:
JEE Details You Should Not Miss:
JEE Coaching Centers: