CBSE Class 11 Properties of Matter and their Measurement Detail & Preparation Downloads
The exploration of the properties of matter stands as a cornerstone of scientific inquiry. Properties such as mass, volume, density, and temperature define the nature of substances and govern their behavior. The measurement of these properties not only unveils the unique characteristics of different materials but also provides a quantitative foundation for scientific investigations. Join us on a journey into the realm of properties of matter and their measurement, where precision and accuracy open the door to a deeper comprehension of the fundamental building blocks of our universe.
A Deep Dive into Properties of Matter with CBSE NCERT Download
Properties Of Matter And Their Measurement
The Fundamentals of Matter: Begin by introducing the basic concepts of matter, emphasizing its role as the substance that occupies space and possesses mass.
Properties that Define Matter: Explore key properties of matter, such as mass, volume, density, and temperature. Explain how these properties provide unique fingerprints for different substances and influence their behavior.
The Art of Measurement: Dive into the world of measurement in chemistry. Discuss the importance of precision and accuracy in obtaining reliable data. Explore common measurement tools and techniques, such as balances, graduated cylinders, and thermometers.
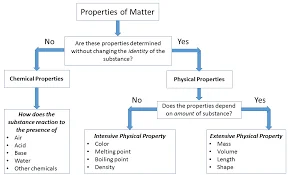
Physical properties
Physical properties refer to characteristics or attributes of a substance that can be observed or measured without altering the substance's composition. These properties provide valuable information about how a material behaves under different conditions. Here's a brief explanation of some common physical properties:
Mass: The amount of matter in an object. It is measured in kilograms or grams.
Volume: The amount of space occupied by an object. It can be measured in cubic meters or cubic centimeters.
Density: The mass of a substance per unit volume. It indicates how tightly packed the particles are within a material.
Color: The visual perception of an object due to the wavelengths of light it reflects or emits.
Odor: The characteristic smell of a substance.
Melting Point: The temperature at which a solid turns into a liquid.
Boiling Point: The temperature at which a liquid turns into a gas.
Solubility: The ability of a substance to dissolve in a solvent, usually water.
Conductivity: The ability of a material to conduct electricity or heat.
Malleability: The ability of a material to deform or flatten under pressure, such as hammering or rolling.
Ductility: The ability of a material to stretch or become elongated when subjected to force, without breaking.
These physical properties help scientists and chemists classify, identify, and understand different substances.
Chemical properties
Chemical properties describe the behavior of a substance as it undergoes chemical reactions or changes in composition. Unlike physical properties, chemical properties involve the interaction of substances with each other. Here's a brief explanation of some common chemical properties:
Flammability: The ability of a substance to burn or ignite in the presence of oxygen. For example, wood is flammable.
Reactivity with Acids and Bases: How a substance reacts with acidic or basic substances. Some materials may produce gases, heat, or other observable changes when exposed to acids or bases.
Corrosiveness: The tendency of a substance to deteriorate or wear away other materials through chemical action. Acids, for instance, are often corrosive.
Toxicity: The degree to which a substance can harm living organisms. Toxic substances can cause adverse effects on health.
Oxidation: The process where a substance reacts with oxygen, often resulting in the formation of oxides. The rusting of iron is an example of oxidation.
Combustibility: Similar to flammability, it describes the ability of a substance to undergo combustion, often producing heat and light.
Radioactivity: The emission of radiation by certain substances, indicating their unstable atomic nuclei. Radioactive elements undergo spontaneous decay.
Chemical Stability: The ability of a substance to resist undergoing chemical changes under specific conditions.
Understanding chemical properties is crucial in predicting how substances will interact with each other in chemical reactions, and it forms the basis for much of the study of chemistry.

CBSE Class 11th Downloadable Resources:
|
1. CBSE Class 11th Topic Wise Summary |
View Page / Download |
|
2. CBSE Class 11th NCERT Books |
View Page / Download |
|
3. CBSE Class 11th NCERT Solutions |
View Page / Download |
|
4. CBSE Class 11th Exemplar |
View Page / Download |
|
5. CBSE Class 11th Sample Papers |
View Page / Download |
|
6. CBSE Class 11th Question Bank |
View Page / Download |
|
7. CBSE Class 11th Topic Wise Revision Notes |
View Page / Download |
|
8. CBSE Class 11th Last Minutes Preparation Resources |
View Page / Download |
|
9. CBSE Class 11th Best Reference Books |
View Page / Download |
|
10. CBSE Class 11th Formula Booklet |
View Page / Download |
Being in CBSE class 11th and considering the board examinations you must be needing resources to excel in your examinations. At TestprepKart we take great pride in providing CBSE class 11th all study resources in downloadable form for you to keep you going.
Below is the list of all CBSE class 11th Downloads available on TestprepKart for both Indian and NRI students preparing for CBSE class 11th in UAE, Oman, Qatar, Kuwait & Bahrain.
SAMPLE PRACTICE QUESTIONS OF SIGNIFICANT FIGURES:
Q1- What are the essential properties of matter?
Answer. The essential properties of matter include mass, volume, density, and the ability to undergo physical and chemical changes.
Q2- How is the mass of an object measured?
Answer. The mass of an object is typically measured using a balance or a scale.
Q3- How is temperature measured?
Answer. Temperature is measured using a thermometer, with common scales being Celsius and Fahrenheit.
Q4- What methods measure the hardness of materials?
Answer. The hardness of materials is often measured using methods like the Mohs scale for minerals or the Rockwell or Vickers scale for metals.
Q5- What is density, and how is it measured?
Answer. Density is the mass per unit volume of a substance and is measured by dividing the mass of an object by its volume. The formula for density is density = mass/volume.

| CBSE CLASS 11th Chemistry Chapters |
| Chapter1: SOME BASIC CONCEPTS OF CHEMISTRY |
| > Importance of Chemistry |
| > Nature of Matter |
| > Uncertainly in Measurement |
| > Laws of Chemical Combinations |
| > Dalton's Atomic Theory |
| > Atomic and molecular Masses |
| > Mole Concept and Molar Masses |
| > Percentage composition |
| > Stoichiometry and Stoichiometric Calculations |
| Chapter2: STRUCTURE OF ATOMS |
| Chapter3: CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES |
| Chapter4: CHEMICAL BONDING AND MOLECULAR STRUCTURE |
| Chapter5: THERMODYNAMICS |
| Chapter6: EQUILIBRIUM |
| Chapter7: REDOX REACTIONS |
| Chapter8: ORGANIC CHEMISTRY - SOME BASIC PRINCIPLE AND TECHNIQUES |
| Chapter9: Hydrocarbons HYDROCARBONS |
| CBSE Class 11 Physics Chapters |
| Chapter1: UNITS AND MEASUREMENTS |
| Chapter2: MOTION IN A STRAIGHT LINE |
| Chapter3: MOTION IN A PLANE |
| Chapter4: LAWS OF MOTION |
| Chapter5: WORK, ENERGY AND POWER |
| Chapter6: SYSTEM OF PARTICLES AND ROTATIONAL MOTION |
| Chapter7: GRAVITATION |
| Chapter8: MECHANICAL PROPERTIES OF SOLIDS |
| Chapter9: MECHANICAL PROPERTIES OF FLUIDS |
| Chapter10: THERMAL PROPERTIES OF MATTER |
| Chapter12: KINETIC THEORY |
| Chapter13: OSCILLATIONS |
| Chapter14: WAVES |
| CBSE Class 11 Mathematics chapter |
| Chapter1: SETS |
| Chapter2: RELATIONS AND FUNCTIONS |
| Chapter3: TRIGONOMETRIC FUNCTIONS |
| Chapter4: COMPLEX NUMBER AND QUADRATIC EQUATIONS |
| Chapter5: LINEAR INEQUALITIES |
| Chapter6: PERMUTATIONS AND COMBINATIONS |
| Chapter7: BINOMIAL THEOREM |
| Chapter8: SEQUENCES AND SERIES |
| Chapter9: STRAIGHT LINES |
| Chapter10: CONIC SECTIONS |
| Chapter11: INTRODUCTION TO THREE-DIMENSIONAL GEOMETRY |
| Chapter12: LIMITS AND DERIVATIVES |
| Chapter13: STATISTICS |
| Chapter14: PROBABILITY |
| Class 8 Link soon |
| Class 9 Link soon |
| Class 10 Link soon |
| Class 12 Link soon |