CBSE Class 11 Quantum Mechanical Model of Atom Detail & Preparation Downloads
The Quantum Mechanical Model of the Atom revolutionized our understanding of the microscopic world, providing a more accurate and comprehensive depiction than its classical predecessors. The quantum Mechanical Model embraces the probabilistic nature of particles. It introduces the concept of wave-particle duality, suggesting that particles, including electrons, exhibit both wave and particle characteristics. Erwin Schrödinger's wave equation lies at the core of this model, describing the probability distribution of finding an electron in a particular region around the nucleus. The model also introduces the idea of quantized energy levels. Electrons are now described as existing in three-dimensional regions known as orbitals, each with a specific energy level. These orbitals have distinct shapes and orientations, contributing to the overall probability of finding an electron in a given space.
Quantum Mechanical Model of the Atom Unveiled in CBSE NCERT Download
Quantum Mechanical Model of Atom
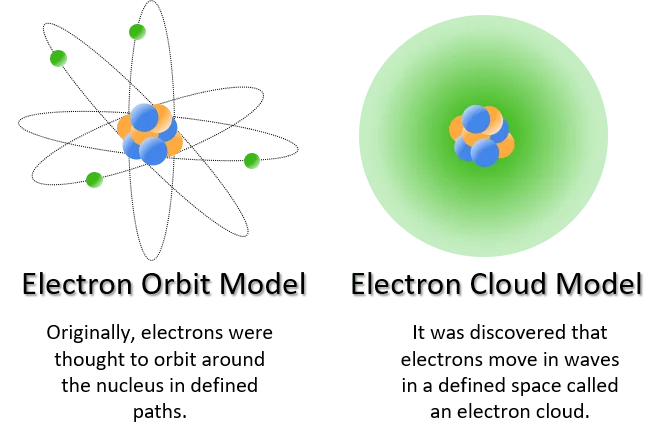
The Quantum Mechanical Model of the Atom is a fundamental framework in quantum physics that describes the behavior of electrons within an atom. It replaced earlier models, such as the Bohr model, which suggested electrons orbit the nucleus in fixed paths. Developed in the early to mid-20th century by scientists like Werner Heisenberg, Erwin Schrödinger, and Max Born, the Quantum Mechanical Model provides a more accurate and comprehensive understanding of atomic structure.
Key features of the Quantum Mechanical Model include:
-
Wave-Particle Duality: Electrons are considered to have both particle-like and wave-like properties. This is a departure from classical physics, which viewed particles strictly as localized entities.
-
Wave Functions: Instead of following definite paths, electrons are described by mathematical functions called wave functions. These functions represent the probability distribution of finding an electron in a particular region around the nucleus.
-
Orbitals: Electrons are found in three-dimensional regions around the nucleus called orbitals. Each orbital is associated with a specific energy level, and they have distinct shapes and orientations. The quantum numbers (principal, azimuthal, magnetic, and spin) characterize these orbitals.
-
Quantized Energy Levels: The energy of electrons is quantized, meaning it exists in discrete levels. Electrons can move between these levels by absorbing or emitting energy in discrete packets known as quanta.
-
Heisenberg Uncertainty Principle: Formulated by Werner Heisenberg, this principle states that it is impossible to simultaneously know with absolute precision both the position and momentum of a particle. This introduces a fundamental limit to our knowledge of the behavior of subatomic particles.
-
Probability Clouds: Rather than depicting fixed electron orbits, the Quantum Mechanical Model represents the likelihood of finding an electron in a particular region around the nucleus. These regions are often visualized as probability clouds.
The Quantum Mechanical Model has withstood extensive experimental testing and is the foundation for understanding the electronic structure of atoms, explaining phenomena such as the periodic table, chemical bonding, and the diverse properties of elements.
Who Discovered the Quantum Mechanical Model?
The development of the Quantum Mechanical Model of the Atom involved the contributions of several physicists in the early to mid-20th century. The key figures associated with the formulation and development of the model include Werner Heisenberg, Erwin Schrödinger, Max Born, and others.
-
Werner Heisenberg (1925): Heisenberg formulated matrix mechanics, a fundamental theory within quantum mechanics, in 1925. He introduced the Heisenberg Uncertainty Principle, highlighting the inherent limitations in simultaneously knowing the precise position and momentum of a particle. This laid the groundwork for a more abstract and mathematical approach to quantum physics.
-
Erwin Schrödinger (1926): Schrödinger developed wave mechanics independently around the same time. He introduced the wave equation that describes the behavior of particles, including electrons, in terms of wave functions. This approach provided a more intuitive and visualizable representation of quantum phenomena, emphasizing the probabilistic nature of particle behavior.
-
Max Born (1926): Born, building on the work of Heisenberg and Schrödinger, interpreted the square of the wave function as a probability density, connecting the mathematical formalism to measurable, observable quantities.
These contributions, along with the insights of other physicists like Louis de Broglie and Paul Dirac, collectively formed the basis of the Quantum Mechanical Model of the Atom.

What is a Quantum Mechanical Orbital?
A quantum mechanical orbital is a three-dimensional region in an atom where there is a high probability of finding an electron. In the Quantum Mechanical Model of the Atom, electrons are not pictured as particles following definite paths around the nucleus, as in the Bohr model. The concept of orbitals arises from the solutions to the Schrödinger wave equation, which is a fundamental equation in quantum mechanics.
Key points about quantum mechanical orbitals include:
-
Three-Dimensional Space: Orbits are defined in three-dimensional space around the atomic nucleus. They describe the regions where electrons are most likely to be found.
-
Quantum Numbers: Each orbital is characterized by a set of quantum numbers—principal (n), azimuthal (l), magnetic (m_l), and spin (m_s). These numbers define the energy level, shape, orientation, and spin of the orbital, respectively.
-
Shapes and Sublevels: Orbitals come in various shapes, and they are often represented as probability density plots or "clouds" where the electron is likely to be located. The principal quantum number determines the energy level and the overall size of the orbital. The azimuthal quantum number determines the shape of the orbital, dividing them into sublevels (s, p, d, f).
- s Orbital: Spherical in shape, with one main sublevel (l = 0).
- p Orbital: Dumbbell-shaped, with three sublevels (l = 1) oriented along the x, y, and z axes.
- d Orbital: More complex shapes, with five sublevels (l = 2).
- f Orbital: Even more complex shapes, with seven sublevels (l = 3).
-
Pauli Exclusion Principle: Each orbital can hold a maximum of two electrons with opposite spins, as dictated by the Pauli Exclusion Principle.
Understanding quantum mechanical orbitals is essential for describing the electronic structure of atoms and molecules.
CBSE Class 11th Downloadable Resources:
|
1. CBSE Class 11th Topic Wise Summary |
View Page / Download |
|
2. CBSE Class 11th NCERT Books |
View Page / Download |
|
3. CBSE Class 11th NCERT Solutions |
View Page / Download |
|
4. CBSE Class 11th Exemplar |
View Page / Download |
|
5. CBSE Class 11th Sample Papers |
View Page / Download |
|
6. CBSE Class 11th Question Bank |
View Page / Download |
|
7. CBSE Class 11th Topic Wise Revision Notes |
View Page / Download |
|
8. CBSE Class 11th Last Minutes Preparation Resources |
View Page / Download |
|
9. CBSE Class 11th Best Reference Books |
View Page / Download |
|
10. CBSE Class 11th Formula Booklet |
View Page / Download |
Being in CBSE class 11th and considering the board examinations you must be needing resources to excel in your examinations. At TestprepKart we take great pride in providing CBSE class 11th all study resources in downloadable form for you to keep you going.
Below is the list of all CBSE class 11th Downloads available on TestprepKart for both Indian and NRI students preparing for CBSE class 11th in UAE, Oman, Qatar, Kuwait & Bahrain.
SAMPLE PRACTICE QUESTIONS OF SIGNIFICANT FIGURES:
Q1. What are quantum mechanical orbitals?
Answer. Quantum mechanical orbitals are three-dimensional regions around the nucleus where there is a high probability of finding an electron. They are characterized by quantum numbers that define the energy, shape, and orientation of the orbital.
Q2. What is the Quantum Mechanical Model of the Atom?
Answer. The Quantum Mechanical Model is a theoretical framework in quantum physics that describes the behavior of electrons within an atom. It abandons the idea of fixed electron orbits and introduces concepts like wave functions and orbitals.
Q3. What are wave functions in the Quantum Mechanical Model?
Answer. Wave functions are mathematical expressions that describe the behavior of particles, including electrons. They provide information about the probability distribution of finding an electron at a particular location in an atom.
Q4. How does the Quantum Mechanical Model differ from the Bohr Model?
Answer. Unlike the Bohr Model, the Quantum Mechanical Model does not depict electrons as particles following fixed paths. Instead, it describes electrons using wave functions and orbitals, emphasizing the probabilistic nature of electron behavior.
Q5. How does the Quantum Mechanical Model differ from the Bohr Model?
Answer. Unlike the Bohr Model, the Quantum Mechanical Model does not depict electrons as particles following fixed paths. Instead, it describes electrons using wave functions and orbitals, emphasizing the probabilistic nature of electron behavior.

| CBSE CLASS 11th Chemistry Chapters |
| Chapter1: SOME BASIC CONCEPTS OF CHEMISTRY |
| Chapter2: STRUCTURE OF ATOMS |
| > Discovery of Sub-Atomic Particle |
| > Atomic Models |
| > Developments Leading to the Bohr’s Model of Atom |
| > Bohr’s Model for Hydrogen Atom |
| > Towards Quantum Mechanical Model of the Atom |
| Chapter3: CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES |
| Chapter4: CHEMICAL BONDING AND MOLECULAR STRUCTURE |
| Chapter5: THERMODYNAMICS |
| Chapter6: EQUILIBRIUM |
| Chapter7: REDOX REACTIONS |
| Chapter8: ORGANIC CHEMISTRY - SOME BASIC PRINCIPLE AND TECHNIQUES |
| Chapter9: Hydrocarbons HYDROCARBONS |
| CBSE Class 11 Physics Chapters |
| Chapter1: UNITS AND MEASUREMENTS |
| Chapter2: MOTION IN A STRAIGHT LINE |
| Chapter3: MOTION IN A PLANE |
| Chapter4: LAWS OF MOTION |
| Chapter5: WORK, ENERGY AND POWER |
| Chapter6: SYSTEM OF PARTICLES AND ROTATIONAL MOTION |
| Chapter7: GRAVITATION |
| Chapter8: MECHANICAL PROPERTIES OF SOLIDS |
| Chapter9: MECHANICAL PROPERTIES OF FLUIDS |
| Chapter10: THERMAL PROPERTIES OF MATTER |
| Chapter12: KINETIC THEORY |
| Chapter13: OSCILLATIONS |
| Chapter14: WAVES |
| CBSE Class 11 Mathematics chapter |
| Chapter1: SETS |
| Chapter2: RELATIONS AND FUNCTIONS |
| Chapter3: TRIGONOMETRIC FUNCTIONS |
| Chapter4: COMPLEX NUMBER AND QUADRATIC EQUATIONS |
| Chapter5: LINEAR INEQUALITIES |
| Chapter6: PERMUTATIONS AND COMBINATIONS |
| Chapter7: BINOMIAL THEOREM |
| Chapter8: SEQUENCES AND SERIES |
| Chapter9: STRAIGHT LINES |
| Chapter10: CONIC SECTIONS |
| Chapter11: INTRODUCTION TO THREE-DIMENSIONAL GEOMETRY |
| Chapter12: LIMITS AND DERIVATIVES |
| Chapter13: STATISTICS |
| Chapter14: PROBABILITY |
| Class 8 Link soon |
| Class 9 Link soon |
| Class 10 Link soon |
| Class 12 Link soon |