CBSE Class 10th Formation and Properties of Ionic Compounds Details & Preparations Downloads
The journey begins with understanding how ions, both positively charged cations and negatively charged anions, come into existence. As we navigate the pages of CBSE NCERT, we'll discover the elemental transformations that set the stage for the mesmerizing dance of electrons, laying the groundwork for the formation of ionic compounds.
Decoding Ionic Compounds: A Comprehensive Guide with CBSE NCERT Download
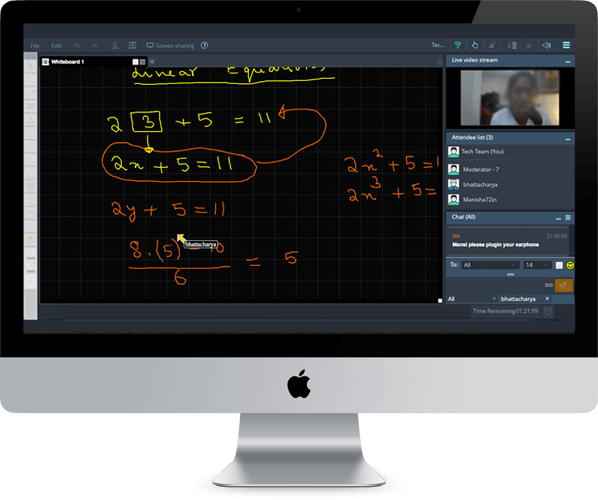
What are Ionic Compounds?
Ionic compounds are a type of chemical compound formed by the combination of positively charged ions (cations) and negatively charged ions (anions). The driving force behind the formation of ionic compounds is the attraction between these oppositely charged ions.
Formation of Ionic Compounds
Step 1: Ion Formation
The process begins with the formation of ions. Metals tend to lose electrons and become positively charged cations, while non-metals gain electrons to become negatively charged anions.
Step 2: Ionic Bonding
The positively charged cations and negatively charged anions are held together by ionic bonds. This electrostatic force of attraction results in the formation of a stable ionic compound.
Properties of Ionic Compounds
Now that we know how they're formed, let's explore some key properties of ionic compounds:
1. High Melting and Boiling Points
Ionic compounds typically have high melting and boiling points. This is because a substantial amount of energy is required to overcome the strong ionic bonds holding the ions together.
2. Solubility in Water
Most ionic compounds are soluble in water. When an ionic compound dissolves, the water molecules surround and separate the ions, allowing them to move freely.
3. Electrical Conductivity
In their solid state, ionic compounds do not conduct electricity because the ions are held in a fixed position. However, when melted or dissolved in water, they become good conductors of electricity as the ions are free to move.
4. Crystalline Structure
Ionic compounds often exhibit a characteristic crystalline structure. The ions arrange themselves in a repeating pattern, forming a crystal lattice.
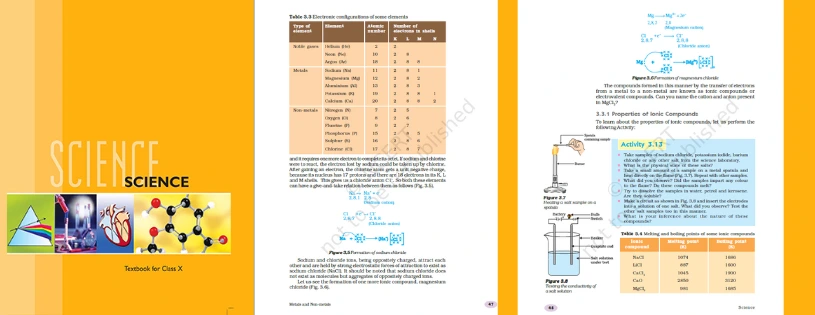
Ionic Compound Properties
1. Physical properties of ionic compounds
Due to the presence of a strong force of attraction between the positive and negative ions, ionic compounds are solids and are hard to break. They generally break into pieces when pressure is applied, hence they are considered brittle.
2. Melting and boiling points of ionic compounds
Due to the presence of electrostatic forces of attraction between ions, a large amount of energy is required to break the ionic bonds between the atoms. Thus, ionic compounds have high melting and boiling points.
3. The solubility of ionic compounds
Ionic compounds are generally soluble in polar solvents such as water whereas solubility tends to decrease in non-polar solvents such as petrol, gasoline, etc.
4. Conduction of Electricity
Ionic compounds do not conduct electricity in the solid state but are good conductors in a molten state. Conduction of electricity involves the flow of charge from one point to another. In the solid state, as the movement of ions is not possible, ionic compounds don’t conduct electricity. Whereas in the molten state, ionic compounds conduct electricity as electrostatic forces of attraction between the ions are overcome by the heat released.
Ionic Character Formula
One way of estimating the ionic character of a bond—that is, the magnitude of the charge separation in a polar covalent bond—is to calculate the difference in electronegativity between the two atoms.
Δχ = χB − χA.
Bond polarity and ionic character increase with an increasing difference in electronegativity.
CBSE Class 10th Downloadable Resources:
| 1. CBSE Class 10th Topic Wise Summary | View Page / Download |
| 2. CBSE Class 10th NCERT Books | View Page / Download |
| 3. CBSE Class 10th NCERT Solutions | View Page / Download |
| 4. CBSE Class 10th Exemplar | View Page / Download |
| 5. CBSE Class 10th Previous Year Papers | View Page / Download |
| 6. CBSE Class 10th Sample Papers | View Page / Download |
| 7. CBSE Class 10th Question Bank | View Page / Download |
| 8. CBSE Class 10th Topic Wise Revision Notes | View Page / Download |
| 9. CBSE Class 10th Last Minutes Preparation Resources (LMP) | View Page / Download |
| 10. CBSE Class 10th Best Reference Books | View Page / Download |
| 11. CBSE Class 10th Formula Booklet | View Page / Download |
Being in CBSE class 10th and considering the board examinations you must be needing resources to excel in your examinations. At TestprepKart we take great pride in providing CBSE class 10th all study resources in downloadable form for you to keep you going.
Below is the list of all CBSE class 10th Downloads available on TestprepKart for both Indian and NRI students preparing for CBSE class 10th in UAE, Oman, Qatar, Kuwait & Bahrain.
SAMPLE PRACTICE QUESTION
Q1: What is an ionic compound?
Ans: An ionic compound is a chemical compound formed by the combination of positively charged ions (cations) and negatively charged ions (anions) through ionic bonding.
Q2: How are ions formed in the context of ionic compounds?
Ans: Ions are formed when metals lose electrons to become positively charged cations, and non-metals gain electrons to become negatively charged anions.
Q3: What is the process of ionic bonding in the formation of ionic compounds?
Ans: Ionic bonding involves the electrostatic attraction between positively charged cations and negatively charged anions, leading to the formation of a stable ionic compound.
Q4: Why do ionic compounds have high melting and boiling points?
Ans: Ionic compounds have high melting and boiling points because a significant amount of energy is required to break the strong ionic bonds holding the ions together in the crystal lattice.
Q5: Are ionic compounds generally soluble in water?
Ans: Yes, most ionic compounds are soluble in water. When dissolved, water molecules surround and separate the ions, allowing them to move freely.

| CBSE CLASS 10 Mathematics Chapters |
| Chapter1: Real Numbers |
| Chapter2: Polynomials |
| Chapter3: Pair of Linear Equations in Two Variables |
| Chapter4: Quadratic Equations |
| Chapter5: Arithmetic Progressions |
| Chapter6: Triangles |
| Chapter7: Coordinate Geometry |
| Chapter8: Introduction to Trigonometry |
| Chapter9: Some Applications of Trigonometry |
| Chapter10: Circles |
| Chapter11: Areas Related to Circles |
| Chapter12: Surface Areas and Volumes |
| Chapter13: Statistics |
| Chapter14: Probability |
| CBSE CLASS 10 Science Chapters |
| Chapter1: Chemical Reactions and Equations |
| Chapter2: Acids, Bases and Salts |
| Chapter3: Metals and Non-metals |
| >Properties of Metals and Non-metals |
| >Reactivity Series |
| >Basic Metallurgical Processes |
| >Corrosion and its Prevention |
| Chapter4: Carbon and its Compounds |
| Chapter5: Life Processes |
| Chapter6: Control and Coordination |
| Chapter7: How do Organisms Reproduce? |
| Chapter8: Heredity |
| Chapter9: Light – Reflection and Refraction |
| Chapter10: The Human Eye and the Colourful World |
| Chapter11: Electricity |
| Chapter12: Magnetic Effects of Electric Current |
| Chapter13: Our Environment |
| Class 8 |
| Class 9 |
| Class 11 |
| Class 12 |