CBSE Class 11 Thermodynamic Terms Detail and Preparation Downloads
Embarking on the captivating realm of thermodynamics opens doors to understanding the intricate dance between energy and matter. Essential thermodynamic terms serve as the compass, guiding us through the fascinating intricacies of systems and their transformations. From the fundamental principles of internal energy and heat exchange to the laws that govern energy conservation, this introduction offers a glimpse into the vocabulary that unlocks the secrets of our universe. Join us as we unravel the core concepts shaping the study of thermodynamics, empowering us to decipher the mysteries of physical processes, energy efficiency, and the very fabric of our reality.
Essential Thermodynamic Terms Unveiled
Dive into the Essence: Unraveling Thermodynamic Mysteries with Key Terms. Explore the fundamental concepts shaping energy transformations and system behavior. From internal energy to entropy, embark on a journey through the core vocabulary that unveils the secrets of our universe's dynamic interplay between matter and energy.
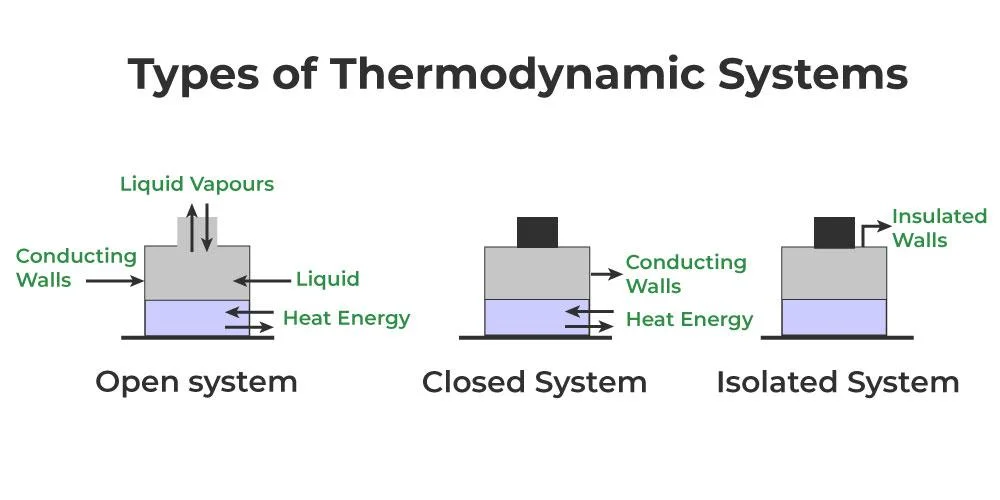
Terms of Thermodynamics
Certainly, I'll provide more detailed explanations for the terms mentioned earlier in the context of thermodynamics:
-
System and Surroundings:
- System: A thermodynamic system is a distinct part of the universe chosen for study. It can be as small as a single molecule or as extensive as the Earth.
- Surroundings: Everything outside the system that interacts with it. The boundaries of the system separate it from the surroundings.
-
Internal Energy (U):
- Internal Energy: This is the sum of all microscopic forms of energy in a system. It includes kinetic energy due to the motion of particles, potential energy due to molecular arrangement, and internal energy changes that can occur due to heat transfer or work done.
-
Work (W) and Heat (Q):
- Work (W): In thermodynamics, work is defined as the energy transferred to or from a system via the application of force along a displacement.
- Heat (Q): Heat is energy transferred between a system and its surroundings due to a temperature difference. It is a form of energy associated with the motion of atoms and molecules.
-
First Law of Thermodynamics:
- First Law: This law of energy conservation states that energy cannot be created or destroyed, only transferred or converted from one form to another. Mathematically, it is expressed as ΔU=Q−W, where is the change in internal energy, Q is heat, and W is work.
-
Enthalpy (H):
- Enthalpy: It is a thermodynamic function that represents the total heat content of a system at constant pressure. It is given by the equation H=U+PV, where P is pressure and V is volume.
-
Entropy (S):
- Entropy: Entropy is a measure of the disorder or randomness in a system. The second law of thermodynamics states that the entropy of an isolated system tends to increase over time.
-
Second Law of Thermodynamics:
- Second Law: This law asserts that the total entropy of an isolated system can never decrease over time. It provides a directionality to natural processes, indicating a tendency toward increased disorder.
-
Thermal Equilibrium:
- Thermal Equilibrium: Two systems are in thermal equilibrium when there is no net transfer of heat between them. Their temperatures are equal, and no further change occurs.
-
Carnot Efficiency:
- Carnot Efficiency: It represents the maximum efficiency any heat engine can achieve operating between two temperature reservoirs. It is determined solely by the temperatures of the reservoirs.
-
Phase Transitions:
- Phase Transitions: These are changes in the state of matter, such as solid to liquid (melting) or liquid to gas (vaporization). The behavior during phase transitions is crucial in understanding material properties.
These terms collectively form the foundation of thermodynamics, enabling the analysis and understanding of energy and matter transformations in various physical systems.

CBSE Class 11th Downloadable Resources:
|
1. CBSE Class 11th Topic Wise Summary |
View Page / Download |
|
2. CBSE Class 11th NCERT Books |
View Page / Download |
|
3. CBSE Class 11th NCERT Solutions |
View Page / Download |
|
4. CBSE Class 11th Exemplar |
View Page / Download |
|
5. CBSE Class 11th Sample Papers |
View Page / Download |
|
6. CBSE Class 11th Question Bank |
View Page / Download |
|
7. CBSE Class 11th Topic Wise Revision Notes |
View Page / Download |
|
8. CBSE Class 11th Last Minutes Preparation Resources |
View Page / Download |
|
9. CBSE Class 11th Best Reference Books |
View Page / Download |
|
10. CBSE Class 11th Formula Booklet |
View Page / Download |
Being in CBSE class 11th and considering the board examinations you must be needing resources to excel in your examinations. At TestprepKart we take great pride in providing CBSE class 11th all study resources in downloadable form for you to keep you going.
Below is the list of all CBSE class 11th Downloads available on TestprepKart for both Indian and NRI students preparing for CBSE class 11th in UAE, Oman, Qatar, Kuwait & Bahrain.
SAMPLE PRACTICE QUESTIONS OF SIGNIFICANT FIGURES:
Q1. What is the First Law of Thermodynamics, and how does it apply to energy conservation?
Answer. The First Law states that energy in a closed system is conserved; it cannot be created or destroyed, only transformed. The change in internal energy (ΔU) is equal to the heat added to the system minus the work done by the system.
Q2. How does entropy relate to the Second Law of Thermodynamics?
Answer. The Second Law states that the total entropy of an isolated system tends to increase over time. Entropy is a measure of disorder or randomness within a system, and its increase reflects the natural direction of processes toward greater randomness.
Q3. What is the significance of Enthalpy (H) in thermodynamics?
Answer. Enthalpy is a measure of a system's total energy content, including internal energy, pressure, and volume. It is particularly useful in constant pressure processes and is often used in heat transfer calculations.
Q4. Can you explain Thermal Equilibrium and its importance?
Answer. Thermal Equilibrium occurs when two systems are at the same temperature, resulting in no net exchange of heat between them. This concept is fundamental for understanding heat transfer and the establishment of thermal balance.
Q5. How does the Carnot Efficiency set a limit for heat engines?
Answer. The Carnot Efficiency represents the maximum efficiency any heat engine can achieve operating between two temperature reservoirs. Real-world engines are always less efficient due to factors like friction and heat loss.

| CBSE CLASS 11th Chemistry Chapters |
| Chapter1: SOME BASIC CONCEPTS OF CHEMISTRY |
| Chapter2: STRUCTURE OF ATOMS |
| Chapter3: CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES |
| Chapter4: CHEMICAL BONDING AND MOLECULAR STRUCTURE |
| Chapter5: THERMODYNAMICS |
| Chapter6: EQUILIBRIUM |
| Chapter7: REDOX REACTIONS |
| Chapter8: ORGANIC CHEMISTRY - SOME BASIC PRINCIPLE AND TECHNIQUES |
| Chapter9: Hydrocarbons HYDROCARBONS |
| CBSE Class 11 Physics Chapters |
| Chapter1: UNITS AND MEASUREMENTS |
| Chapter2: MOTION IN A STRAIGHT LINE |
| Chapter3: MOTION IN A PLANE |
| Chapter4: LAWS OF MOTION |
| Chapter5: WORK, ENERGY AND POWER |
| Chapter6: SYSTEM OF PARTICLES AND ROTATIONAL MOTION |
| Chapter7: GRAVITATION |
| Chapter8: MECHANICAL PROPERTIES OF SOLIDS |
| Chapter9: MECHANICAL PROPERTIES OF FLUIDS |
| Chapter10: THERMAL PROPERTIES OF MATTER |
| Chapter12: KINETIC THEORY |
| Chapter13: OSCILLATIONS |
| Chapter14: WAVES |
| CBSE Class 11 Mathematics chapter |
| Chapter1: SETS |
| Chapter2: RELATIONS AND FUNCTIONS |
| Chapter3: TRIGONOMETRIC FUNCTIONS |
| Chapter4: COMPLEX NUMBER AND QUADRATIC EQUATIONS |
| Chapter5: LINEAR INEQUALITIES |
| Chapter6: PERMUTATIONS AND COMBINATIONS |
| Chapter7: BINOMIAL THEOREM |
| Chapter8: SEQUENCES AND SERIES |
| Chapter9: STRAIGHT LINES |
| Chapter10: CONIC SECTIONS |
| Chapter11: INTRODUCTION TO THREE-DIMENSIONAL GEOMETRY |
| Chapter12: LIMITS AND DERIVATIVES |
| Chapter13: STATISTICS |
| Chapter14: PROBABILITY |
| Class 8 Link soon |
| Class 9 Link soon |
| Class 10 Link soon |
| Class 12 Link soon |