The Complete IB Chemistry Study Guide: SL and HL
The IB Chemistry Guide and Notes for SL and HL to fetch you better scorers in the IB test is not going to be skyrocketing. But scrutiny would take you places. IB Chemistry is not too hard or too simple, but it all depends on how you look at it. More like it is how best you get hold of the syllabus and the study guide you are getting. We have crafted the best IB Chemistry Study Guide for SL and HL students. Get Free IB Chemistry Trial Session Download IB Chemistry eBook Download IB Chemistry Guide Here Download IB Chemistry Syllabus Here Paper 1 Paper 2 Paper 3 (Options) IA Standard Level (SL) 30 multiple choice questions 45 min 20% Short answers 1 hr 15 min 40% Short answers 1 hr 20% 20% Higher Level (HL) 40 multiple choice questions 1 hr 20% Short answers 2 hr 15 min 36% Short answers 1 hr 15 min 24% 20% Periodic table only (supplied) No calculators Calculators and Data Booklet Day 1- Paper 1 (30 MCQ / 45 Minumtes) Mark Range 0-7 8-12 13-18 19-20 21-23 24-26 27-30 Grade 1 2 3 4 5 6 7 Paper 2 (Short Answers/ 1 Hour 50 Minutes) Mark Range 0-7 8-12 13-18 19-20 21-23 24-26 27-30 Grade 1 2 3 4 5 6 7 Day 2- Paper 3 (Options / 1 Hour) Mark Range 0-7 8-12 13-18 19-20 21-23 24-26 27-30 Grade 1 2 3 4 5 6 7 Day 1- Paper 1 (40 MCQ / 1 Hour) Mark Range 0-7 8-12 13-18 19-20 21-23 24-26 27-30 Grade 1 2 3 4 5 6 7 Paper 2 (Short Answers/ 2 Hour 15 Minutes) Mark Range 0-7 8-12 13-18 19-20 21-23 24-26 27-30 Grade 1 2 3 4 5 6 7 Day 2- Paper 3 (Options / 1 Hour 15 Minutes) Mark Range 0-7 8-12 13-18 19-20 21-23 24-26 27-30 Grade 1 2 3 4 5 6 7 Pressure at STP redefined in data booklet: STP conditions = 273 K and 100 kPa; SATP conditions = 298 K and 100 kPa. Molar volume of an ideal gas at STP = 2.27 × 10−2 m3 mol−1 (= 22.7 dm3 mol−1) 4.3 Electron domains now used in place of negative charge centres. Coordinate bond exclusively used in place of dative bond 4.4 London (dispersion) forces used for instantaneous induced dipole-induced dipole forces of attraction; Van der Waals’ forces redefined as an inclusive term to include dipole-dipole, dipole induced dipole and London (dispersion) forces 8.1 Amphiprotic species can act as Bronsted--‐Lowry acids and bases; amphoteric substances can act as acids and bases, including reactions where protons are not transferred. Topic 9 : Oxidation and reduction 9.1 Oxidation state uses Arabic numerals e.g. +2, ‐3; oxidation number uses Roman numerals in the nomenclature of a compound e.g. Fe(II)O. 9.2 Definition of electrochemical cell is now an inclusive term to include voltaic and electrolytic cells. 10.1 Functional group refers to the part of a molecule responsible for specific properties e.g. –OH hydroxyl; class of compound refers to the family of compounds/homologous series e.g. alcohols. 13.1 Transition element redefined to include scandium. 18.3 pH curves now used in place of titration curves. 20.3 Cis-trans and E/Z isomerism used in place of geometric isomerism The Group 4 Project is a compulsory activity that all IB Chemistry Diploma Programme students must be a part of. The main agenda of this assessment is to understand and analyse a Chemistry related issue and problem. This activity emphasises on the process and knowledge rather than "The Assessment" Internal Assessment is one of the most important part of IB Diploma Chemistry course, which is compulsory for both Higher Level and Standard Level students. It helps students to exhibit the application of their skills and knowledge. The requirements and guidlines for both SL and HL students are the same. It contributes 20% to the final assessment. Download IB Chemistry IAs Student Support Guide Investigation Title Student Work Annotated 1 2 3 4 5 Impact of increased water acidity on coral reefs based on a study of the kinetics of CaCO3 and HCl 6 A study of the saponification reaction of olive oil 7 8 9 10 11 Hydrolysis of PVA 12 Ion absorption properties of sodium polyacrylate 13 14 15 Boiling points of binary mixtures 16 17 Oxidation of luminol 18 Duration: 10 Hours Weightage: 20% Download IB Chemistry IA Scoring Guide Here Both IB Chemistry SL and HL will cover the 11 topics in the order with same subtopics listed in the following IB Chemistry Guide discussions. (The time allotted in the bracket is only indicative). Click on the links to find the respective Notes 1.1. Particulate Nature of Matter and Chemical Change Key IB points to keep in mind Key IB points to keep in mind Notes – Mole Concept and Avogadro’s Constant 1.3 Reacting Masses and Volumes Key IB points to keep in mind - Notes – Mass & Gaseous Volume Relationships in Chemical Reactions Notes – All of the Stoichiometry 2.1 The Nuclear Atom Key IB points to keep in mind: Key IB Points to keep in mind: Key IB points to keep in mind 3.1 Periodic Table 3.2 Periodic Trends Key IB points to keep in mind: 4.1 Ionic Bonding and Structure 4.2 Covalent Bonding Key IB points to keep in mind Key IB points to keep in mind Key IB points to keep in mind: 4.5 Metallic Bonding Key IB points to keep in mind Notes - Energetics Key IB points to keep in mind 5.2 Hess’s Law Key IB points to keep in mind 5.3 Bond Enthalpies Key IB points to keep in mind Get Free IB Chemistry Trial Session Download IB Chemistry eBook 6.1 Collision Theory and Rates of Reaction Key IB points to keep in mind 6.2 Notes – Collision theory 6.3 Notes – Rates of Reaction 7.1 Equilibrium Key IB points to keep in mind Notes – The position of Equilibrium Study Guide – Equilibrium Key IB points to keep in mind 8.2 Properties of Acids and Bases Key IB points to keep in mind Study Guide – Acids and Bases 8.3 The pH Scale Key IB points to keep in mind 8.4 Strong and Weak Acids and Bases Key IB points to keep in mind Notes – Strong and Weak Acids and Bases 8.5 Acid Deposition Key IB points to keep in mind Key IB points to keep in mind Notes – Oxidation and Reduction Study Guide – Oxidation and Reduction Key IB points to keep in mindThe Complete IB Chemistry Study Guide for SL & HL 2021
This IB Chemistry Guide includes:
IB Chemistry Guide Book Download


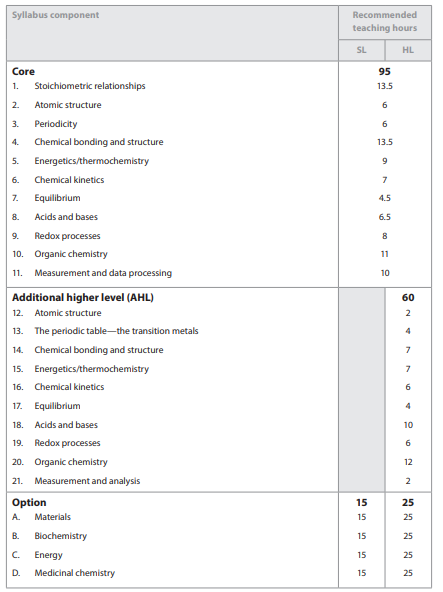
IB Chemistry Syllabus Guide
IB Chemistry Resources
IB Chemistry Exam Overview Guide
IB Chemistry Exam Scoring Guide
Standard Level
Higher Level
IB Chemistry Calculators Guide
IB Chemistry New Terminology Guide
Topic 1: Stoichiometric relationships 1.3
Topic 4 : Bonding
Topic 8 : Acids and bases
Topic 10 : Organic chemistry
Topic 13 : The periodic table - the transition metals
Topic 18 : Acids and bases
Topic 20 : Organic chemistry
IB Chemistry Group 4 Project Guide
IB Chemistry IA Overview Guide
IB Chemistry Internal Assessment (IA) Student Support Guide



IB Chemistry Internal Assessment Examples
Factors effecting the boiling and melting points in organic homologous series
The effect of halogen atom substitution on bond angles in halogenated compounds
Positive inductive effect of methyl groups in nine simple alcohols
An investigation into the dependence of egg protein denaturation on temperature
Investigating the oxidative rancidity of polyunsaturated oils
The enthalpy of combustion of alcohols
Investigating lipids suitable for manufacturing soap for a school project
Effect of cooking time on chlorophyll degradation
Catalysis of iodine clock reaction
Lattice enthalpy
Effect of red wine tannin concentration on methylene blue oxidation
Reaction kinetics of hydrolysis of sucrose using polarimetry
IB Chemistry IA Scoring Guide
Internal Assessment Criteria
Personal Engagement
Exploration
Analysis
Evaluation
Communication
Total
2 (8%)
6 (25%)
6 (25%)
6 (25%)
4 (17%)
24 (100%)
IB Chemistry Notes for SL and HL
Topic 1: Stoichiometric Relationships - 13.5 Hours for SL and HL
Topic 2: Atomic Structure - 6 Hours for SL and HL
Topic 3: Periodicity - 6 Hours for SL and HL
Topic 4: Chemical Bonding and Structure - 13.5 Hours for SL and HL
Topic 5: Energetics/Thermochemistry - 9 Hours for SL and HL
Topic 6: Chemical Kinetics - 7 Hours for Both SL and HL
Topic 7: Equilibrium - 4.5 Hours for SL and HL
Topic 8: Acids and Bases - 6.5 Hours for SL and HL
8.1 Theories of Acids and Bases
Topic 9: Redox Processes - 8 Hours for SL and HL









 Chemistry Study Guide.png)